🫀 Secrets of the human vessel
How a 2004 SARS Lab Cluster Changed Biosafety in Beijing
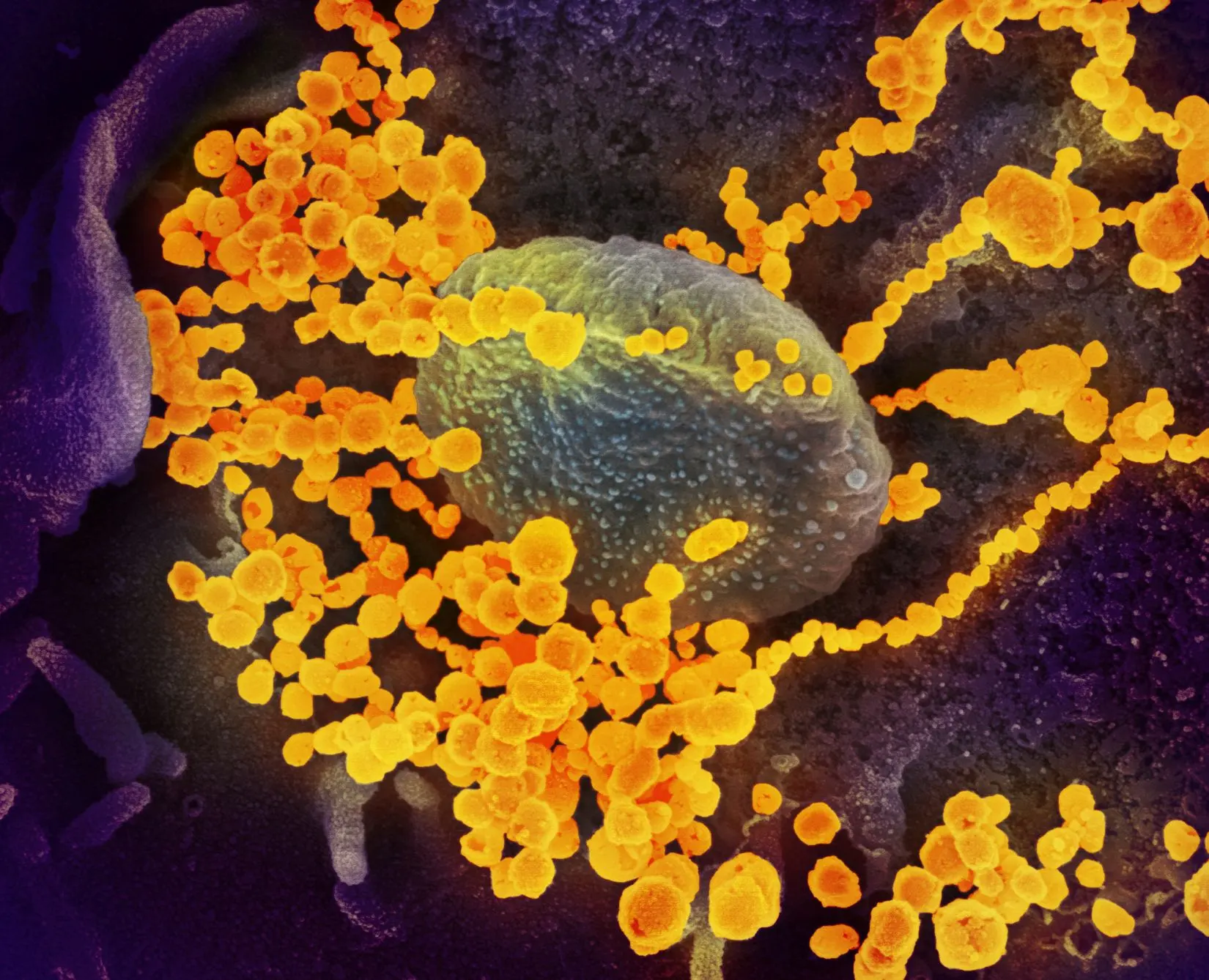
- What: A 2004 cluster of SARS infections in Beijing was linked to laboratory work, highlighting biosafety failures and prompting stronger laboratory safety measures in China.
- Where: Beijing, China.
- When: 2004, after the original SARS outbreak had receded.
In 2004, a small cluster of SARS infections in Beijing did not begin in a market, a hospital ward, or a household. It was linked to laboratory work.
WHO Investigation Into the Beijing Cluster
According to the World Health Organization, staff at a virology institute handling SARS coronavirus samples became infected, raising the immediate possibility of further transmission beyond the lab. The event was limited, but it exposed a problem with wider consequences: even after the original SARS outbreak had receded, the virus still posed a risk in research settings.
Biosafety Weaknesses in Laboratory Work
The significance of the incident was not only that infections occurred, but that they pointed to weaknesses in biosafety practice. Handling dangerous pathogens depends on routine controls being followed exactly, from training and supervision to containment procedures. When one part fails, the breach is no longer just an occupational hazard. It becomes a public health issue.
Stronger Biosafety Measures in China
That is why the Beijing cases mattered beyond the number of people directly affected. They renewed attention on how high-risk samples were stored, processed, and studied, and whether existing safeguards were being applied consistently enough. In practical terms, the episode helped drive stronger biosafety measures in Chinese virology laboratories.
The larger lesson was concrete rather than dramatic. Outbreak prevention is not only about finding new viruses in the wild. It also depends on preventing accidental exposure in the places meant to study them safely. The 2004 SARS lab infections became an early warning that containment standards inside laboratories can shape risks outside them.
Did You Know?
The WHO later identified multiple laboratory-associated SARS incidents after the main epidemic had ended, making biosafety lapses a major concern.